Lignin is the key material that confers rigidity to the walls of cells and tissues of most plants. It is the largest untapped renewable source of raw materials and is made of many of the same chemical building blocks as those found in petroleum-based materials; and has great potential as a raw material as an alternative to petroleum in the production of many essential compounds we use in our daily lives.
Effective conversion of lignin involves finding ways to increase the production of bio-oil and reduce a solid by-product called “char.” Recent studies suggest that pretreatment is necessary to improve product yields and lower char formation. However, there is limited understanding of the effects of various pretreatment parameters on lignin conversion.
Now, research by a team of Department of Chemical and Biological Engineering scientists, led by associate professor and Richard C. Seagrave Professor Jean-Phillippe Tessonnier, is bridging this gap by characterizing chemical transformations occurring during lignin pretreatments, and the catalytic parameters that govern the formation of chemical building blocks from the pretreated product. “An effective strategy to produce highly amenable cellulose and enhance lignin upgrading to aromatic and olefinic hydrocarbons” has been published in the Royal Society of Chemistry journal Energy & Environmental Science.
Co-authors of the research include Daniel Vincent Sahayaraj and Hamed Bateni (former Tessonnier doctoral students), A Lusi and Harish Radhakrishnan (former and current Mechanical Engineering doctoral students, respectively), Alireza Saraeian and Andew J. Kohler (former and current Chemical Engineering doctoral students, respectively); Brent H. Shanks (Anson Marston Distinguished Professor in Engineering, Mike and Jean Steffenson Chair and director of the Center for Biorenewable Chemicals); and Xianglan Bai (associate professor, Department of Mechanical Engineering).

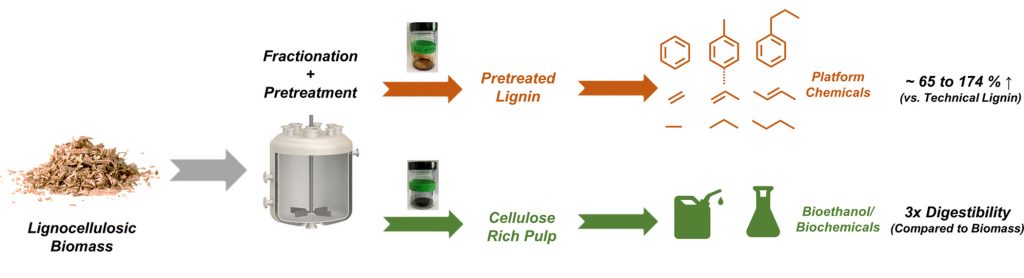
The researchers found that compared to lignin converted using conventional methods, their technique increased production of the desired compounds by up to 174%, in a substantially cost-effective manner. Furthermore, the residual pulp obtained using this method was enriched in sugars and was thus more amenable for conversion to fuels and chemicals than the parent biomass.
The breakthrough comes from the discovery that supercritical ethanol combined with a metal catalyst effectively lower char formation and enhance the volatilization of lignin-derived carbon. The strategy was then extended directly to lignocellulosic biomass (corn stover, switchgrass, red oak) to fractionate and pretreat lignin in a one-pot approach. The lignin oil obtained from this process exhibited an excellent potential to be converted into platform chemicals.
Overall, this work is seen as paving the way for production of greener chemicals and biofuels through efficient lignin conversion, for a better future.